(Image via SQI Diagnostics Inc.)
“Integrating rapid diagnostics is a major step forward in lung transplantation. By providing transplant teams with quantitative metrics to more accurately assess donor lungs, we are moving decision making in transplantation into the era of personalized medicine.”
- Dr. Shaf Keshavjee, Surgeon-in-Chief, Sprott Department of Surgery, Director, Toronto Lung Transplant Program and Director, Latner Thoracic Surgery Research Laboratories.

The costs associated with anything in the medical field are high, this we know, but costs are always highest where there are unmet needs, and where this occurs, we find opportunity.
According to the Canadian Institute for Health Information, there are roughly 15,000 donor lungs available in North America yearly – but only about 3,000 (20%) are transplanted. For those that do receive a transplant, there is also the issue of debt, life at risk post-op, hospitalization is expensive, but a unique company in the med-tech space is out to fill this gap and help people breathe a little easier, perhaps it could spell good news for an investment portfolio.
SQI Diagnostics are leaders in the science of lung health. We develop and manufacture respiratory health and precision medicine tests that run on SQI’s fully automated systems. We’re driven to create and market life-saving testing technologies that help more people in more places live longer, healthier lives. The firm is focused on developing the business units targeting organ transplant, autoimmune disease, and serological testing.
Out to be leaders in the science of lung health, SQI Diagnostics Inc. (TSX-V: SQD, OTCQB: SQIDF, Forum) develops and manufactures respiratory health and precision medicine tests that run on its automated systems.
SQI’s tests simplify and improve COVID19 antibody monitoring, Rapid Acute Lung Injury testing, donor organ transplant informatics, and immunological protein and antibody testing. Its business involves in commercializing its developmental COVID-19 antibody test kit, and the RALI-Dx and RALI-fast Point-of-Care tests to triage COVID-19 patients for respiratory distress.

(Image via SQI Diagnostics Inc. click to enlarge.)
The company was founded in 1999 and is headquartered in Toronto, Canada and its geographical segments include Canada, Europe, and the United States, where the company earns more than half of its revenues.
About the company:
This precision medicine company is unique in its space for its work with rapid lung donor and other respiratory diagnostic testing products and testing services for healthcare providers, patients, and North American consumers.
SQI Diagnostics also offers TOR-dx, a lung test, which enables transplant surgeons to assess the health of a donor's lung; RALI-Dx, a COVID-19 severity triage lab test for COVID-19 positive patients in the hospital the; RALI-fast, a COVID-19 severity triage POC test and a COVID-19 at-home antibody test kit.. The Company also distributes a range of COVID-19 rapid, mobile PCR and antigen tests in Canada.
SQI Diagnostics’ rapid diagnostic tests are sold to healthcare professionals so that patients can get accurate results and fast effective treatment, and direct-to-consumers so that individuals can take action to improve their health outcomes from the comfort of their home.
Under organ transplant, SQI is pioneering the development of an advanced diagnostic `the donor organ prior to transplant surgery and that makes available viable donor organs that would have otherwise been rejected during transplant. .
The company’s TORdx Lung Test detects inflammation at the molecular level enabling surgeons to make a “go” or “no-go” decision on initiating transplantation; there is currently no other such test. SQI has partnered with University Health Network (UHN) Hospitals, one of the largest health and medical research organization in North America, for the TORdx Lung Test clinical development program and the most active lung transplant hospital in the world – they are the key opinion leaders globally for advances in lung transplant medicine. SQI is working with agencies in both the US and Canada to submit the TORdx Lung Test for regulatory approval.
Additional diagnostic tests designed to increase the chance of successful organ transplant for the kidney and liver are planned for clinical development post-regulatory approval of the TORdx Lung Test.
Looking at its autoimmune disease testing, SQI has direct-to-consumer Celiac Disease and a Rheumatoid Arthritis (RA) Tests that enable people to screen for the diseases from the comfort of their home. The direct-to-consumer RA Test can help identify and confirm RA symptoms for early care. The direct-to-consumer Celiac Test is used by consumers to confirm that they have Celiac biomarkers and they can also validate the effectiveness of dietary and lifestyle changes to confirm the autoimmune response is improving.

(Image via SQI Diagnostics Inc. click to enlarge.)
In its other direct to consumer testing, SQI is has launched its direct-to-consumer EXACT COVID-19 Antibody Test to detect the presence of SARS-CoV-2 antibodies in the blood. The EXACT COVID-19 Antibody Test will determine if someone has been exposed to the SARS-CoV-2 virus and the degree of exposure and possible immunity to it and will also measure antibodies produced through infection with COVID-19. SQI’s EXACT COVID-19 Antibody Test Kit’s accuracy exceeds 99% The EXACT COVID-19 Antibody test is approved by the New York State Department of Health (“NYSDOH”). It is initially being sold through its US-based marketing partner AZOVA Inc., and was approved through and will be run at its New York State-based CLIA lab testing partner KSL Diagnostics Inc. The Company can market, sell and collect samples anywhere in the US – the samples must be run in its NYSDOH approved and CLIA licesnsed lanb in New York State.
This test could be the first, or one of the first, antibody tests shipped direct to the consumer home or business. People won’t have to travel to a clinic or hospital to be tested for the presence of SARS-CoV-2 antibody. The test is also unique in that the test’s blood sample is obtained through a simple finger stick, it measures six important COVID-19 antibodies and can be used to monitor changes to an individual’s COVID-19 antibody levels from vaccination or infection over time allowing them to monitor possible changes in their immune (protection) status.
SQI is also fast-tracking the development of a COVID-19 Triage Test called the RALI-Dx (il-6). The RALI-Dx test is an Inflammatory response test comprised of several biomarkers indicative of the “cytokine storm” that is associated with acute lung injury.
The Rapid Acute Lung Injury Diagnostic Test (“RALI-Dx (il-6) ”) test can deliver results in under an hour, giving the clinician insight into whether a patient with COVID-19, Flu, or any upper respiratory condition leading to Acute Lung Injury, can be treated and released for home care, should be admitted to a standard hospital unit to be monitored, or is likely to develop severe life-threatening symptoms and requires immediate intensive care unit (ICU) admittance and advanced respiratory care measures. The ability to effectively triage COVID-19 (or Flu) patients for onset of Rapid Acute Lung Injury would be a diagnostic industry first. The company has the RALI-dx (il-6) test in front of Health Canada seeking interim order approval to aid doctors in their assessment of COVID19 patients. The company is also developing an expanded RALI-dx test with multiple biomarkers that is targeted beyond COVID19 patients and at the broader market of all patients entering the hospital with Rapid Acute Lung Injury symptoms.
CEO Morris explained that the test predicts the probability that a COVID positive patient in this case would end up intubated in the ICU.
“The test will then show the clinician or the emergency doctor, who could triage somebody to home, or just admit to a general medicine ward for observation, or treat more aggressively with the goal of avoiding intubation and mechanical ventilation in the ICU, after which, historically the mortality and morbidity for COVID patients is quite a bit higher”
In the news:
In March 2022, SQI Diagnostics’ partner, KSL Diagnostics has received conditional approval from the New York State Department of Health for the EXACT COVID-19 Antibody Test. Access to the EXACT COVID-19 Antibody Test is now be available to physicians and their patients in the United States through AZOVA Inc.
SQI Diagnostics CEO Andrew Morris sat down with Shoran Devi to discuss the news.
(Click to play.)
“The EXACT COVID-19 Antibody Test is the next generation of tools in the fight against COVID-19. It gives peace of mind because it can detect an immune response in both vaccinated and previously COVID-19 infected people. It also has the potential to give the health care system key insights and data into the relationship between antibody levels and breakthrough infections and reinfections. We’re bringing these tests to market now because we are determined to do our part to end the pandemic.”
CEO Morris clarified to Stockhouse Editorial in an interview that approval in New York state means that through this approval, it can be marketed by SQI anywhere in the United States.
“We can collect samples from customers anywhere in the US. The test just simply has to be run in the approved lab in New York state.”
Serving as the company’s marketing partner, AZOVA is a national organization that has 5,000 points of presence in the US in its clinical and testing network. New York being a hotbed of COVID activity for years means that this could lead to an immense database from the information that is gathered.
Looking ahead:
The team sees several catalyst evens on the horizon. As soon as mid-April, the company is launching its EXACT COVID-19 Antibody Test and expect to start delivering revenues from that in calendar Q2 2022. A RALI-Dx product is in front of Health Canada, and it is expected that there will be a positive outcome from that in Q2.
The team is also working to complete last bit of clinical work on the TORdx Lung Test so that it can be filed with the FDA and Health Canada, anticipated to be a H2 2022 milestone.
As the company expands its clinical work, CEO Morris noted to Stockhouse Editorial that they could pick up more TORdx customers along the way.
Clinical Development Progress 4 Innovative Diagnostic Products Moving Through Clinical / Regulatory:
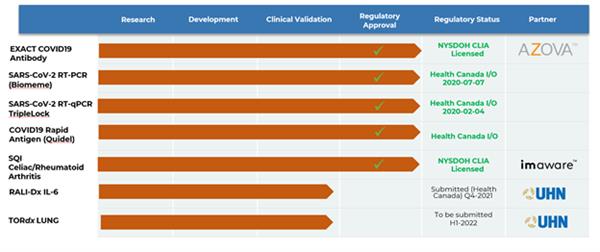
(Image via SQI Diagnostics Inc. click to enlarge.)
The company has partnered with Owlstone Medical to expand its Lung Transplant testing portfolio to non-invasive post-transplant monitoring tests. Two products are targeted to arise from the partnership. The first is a test to detect a fungus that affects over 55% of post-transplant patients. The tests are unique in that they are non-invasive and use breath-based sample collection instead of the current standard of care – a bronchiolar scope and fluid or tissue sample collection and Owlstone is currently completing a clinical study in the Netherlands to further support the product performance.
A leader in breath biopsy for applications in early disease detection and precision medicine, Owlstone Medical is out to save 100,000 lives by realizing the enormous promise of breath-based diagnostics. Owlstone Medical is conducting independent clinical trials to validate breath-based volatile organic compounds to diagnose Invasive Aspergillosis with high levels of performance.
SQI and Owlstone Medical will agree on the most appropriate regulatory strategy for the tests and can seek approval of the breath collector as a medical device and / or the approval of the lung transplant rejection in vitro diagnostic test, with the appropriate regulatory authorities.
Meet the team:
Public since 2007, the company is led by a team of experienced professionals who have seen great changes in the medical landscape.
CEO Morris helped take the business public and has more than 20 years life sciences industry experience, having been Managing Partner ATWILL Medical Solutions, as well as Co-Founder and CFO of Planet People (Concrobium products – sold to Rustoleum for $80 million in 2018, and he was a Co-founder of FORGE Hydrocarbons, developer of a Lipid-to-Hydrocarbon technology that produces “drop in” renewable fuels that are indistinguishable from petroleum-derived fuels. Morris served in the Canadian Forces for 13 years, worked for several technology start-ups, and Scotia Capital in equity research.
He is joined by CSO Dr. Eric Brouwer, who also serves as CSO of Trinity Biotech plc, and was CEO of Fiomi Biotech, and Director of Assay R&D at Abbott Diagnostics, along with Senior Scientist at the point- of care diagnostic innovator i-STAT (acquired by Abbott Diagnostics).
Morlan Reddock, the team’s CFO was Director of Finance for Greater Toronto Airport Authority and his resume includes VP Finance for Ronnex, Distribution Director of Finance Treasury and Investor Relations for SiriusXM Canada.
Investment summary:

(1: This represents a blended average of management's internal estimates, ED visits for Respiratory Distress in Canada and the US : NACRS Emergency Department Visits and Length of Stay by Province/Territory, 2019–2020 & National Hospital Ambulatory Medical Care Survey: 2018 Emergency Department Summary Tables (cdc.gov) & Based on internal estimates, 2019 transplant data (Global Observatory on Donation and Transplantation) and Waitlist data (Canadian Blood Services National Organ Waitlist 2012-2018 & Health resources and Services Administration – www.organdonor.gov)
2: https://www.medtechdive.com/news/as-antibody-tests-proliferate-analysts-see-multibillion-dollar-market-for/576408/, Based on internal estimates, ED visits for Respiratory Distress in Canada and the US : NACRS Emergency Department Visits and Length of Stay by Province/Territory, 2019–2020 & National Hospital Ambulatory Medical Care Survey: 2018 Emergency Department Summary Tables (cdc.gov), Based on internal estimates, 2019 transplant data (Global Observatory on Donation and Transplantation) and Waitlist data (Canadian Blood Services National Organ Waitlist 2012-2018 & Health resources and Services Administration – www.organdonor.gov) Rheumatoid Arthritis Diagnosis Tests- Global Market Trajectory & Analytics, Global Industry Analytics Inc. Image via SQI Diagnostics Inc. click to enlarge.)
With its platforms awaiting approval from Health Canada and the FDA, SQI Diagnostics continues to advance its pipeline of market-ready and late-stage products, gaining upside exposure to COVID-19 testing.
Boasting State-of-the-art international organizational standard manufacturing facility and capabilities, its donor lung transplant products anchor its respiratory health pipeline.
Driven to create and market lifesaving testing technologies that help more people in more places live longer, healthier lives, SQI Diagnostics gives medical practitioners and patients powerful all-in-one-well efficiencies while generating gold-standard data.
For more information, please visit sqidiagnostics.com.
FULL DISCLOSURE: This is a paid article produced by Stockhouse Publishing.