ASCO has informed the Company that the previously accepted MDNA11 oral abstract has been withdrawn due to alleged violation of their prior publication policy based solely on their review of the abstract presented at AACR 2024.
Medicenna disagrees with ASCO’s decision as the AACR 2024 abstract was limited to on-going interim results whereas the ASCO 2024 abstract specified data from a completed phase 1 study that were not presented at AACR 2024.
No other matters were raised by ASCO regarding any data included in the submitted abstract, the planned data intended for presentation at ASCO, or any aspect of the clinical trial.
The Company looks forward to sharing the full high-impact data set of our potentially best-in-class IL-2 and emerging results from the ABILITY-1 trial of MDNA11 at a special virtual event to be organized over the next 2 to 4 weeks. Details on the event will follow.
TORONTO and HOUSTON, May 13, 2024 (GLOBE NEWSWIRE) -- Medicenna Therapeutics Corp. (“Medicenna” or the “Company”) (TSX: MDNA, OTCQB: MDNAF), a clinical-stage immunotherapy company focused on the development of Superkines, announced today that the American Society of Clinical Oncology ("ASCO") decided to reverse its decision and to withdraw Medicenna’s abstract entitled “Results from ABILITY-1 Monotherapy Dose Escalation Study with MDNA11, an Engineered Long-acting IL-2 agonist, in patients with advanced solid tumors,” which had been initially selected for an oral podium presentation at ASCO’s 2024 Annual Meeting. ASCO advised the Company that the reason for this withdrawal was that the Program Committee had determined that the Company had allegedly violated ASCO’s “Prior Publication Policy” by previously reporting results at the 2024 Annual Meeting of the American Association of Cancer Research (“AACR”). No other concerns were raised by the ASCO Program Committee.
The Company disagrees with the ASCO Program Committee’s decision to withdraw the oral podium presentation. In the Company’s opinion, it did not violate ASCO’s Prior Publication Policy. While the Company did present results at the AACR, the AACR abstract explicitly referenced a data cut-off date of December 23, 2023, and only on-going interim monotherapy results were presented, whereas the ASCO abstract specified that the results presented would focus on a complete Phase 1 monotherapy data set without a data cut-off date. This complete data set was analyzed and prepared specifically for presentation at ASCO 2024.
“Medicenna is disappointed by ASCO’s Scientific Program Committee’s decision, as we are now unable to share our ground-breaking results that demonstrate ongoing deep and durable responses in advanced solid tumors as a single agent in the ABILITY-1 clinical trial of MDNA11 at ASCO,” said Dr Fahar Merchant, President and CEO of Medicenna. “The potential to present data from the ABILITY-1 trial at ASCO would have been a tremendous honor, reflecting the investment of many patients, their families and investigators in the trial that has demonstrated promising advancement in cancer care, bringing comfort of a potential cure to patients through development of this highly promising molecule. The ABILITY-1 trial demonstrates robust single agent activity in tumor types not previously shown to have durable response to IL-2 based therapies—a significant feat. Given the strong interest by many stakeholders in our MDNA11 program, we look forward to sharing the full high impact data set at a special virtual event to be organized over the next 2 to 4 weeks with more details to be announced shortly.”
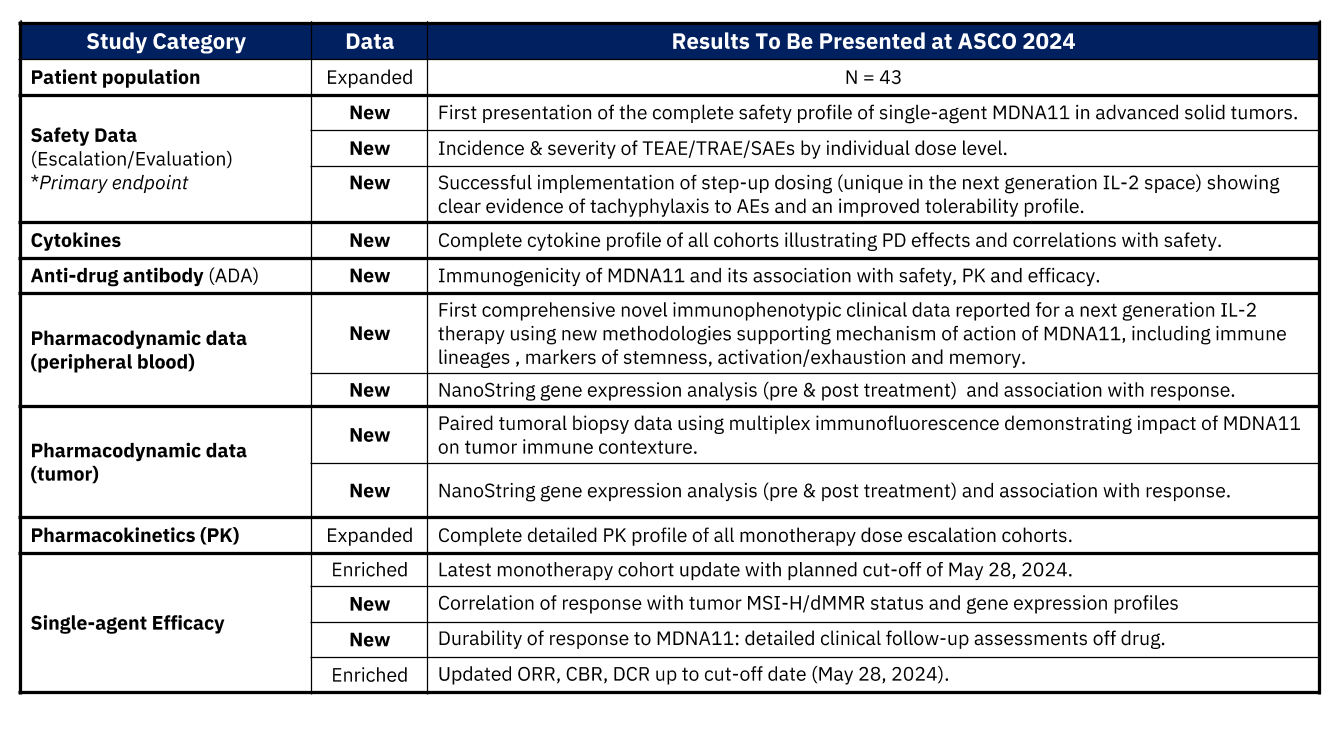
At the event, clinical data originally planned for the 2024 ASCO Annual Meeting and that has not previously been presented at any external conference, including AACR (summarized in the table below), will be presented from the ongoing Phase 1/2 ABILITY-1 Study evaluating MDNA11, a long-acting ‘beta-enhanced not-alpha’ interleukin-2 (IL-2) super-agonist, as both a monotherapy and in combination with pembrolizumab (KEYTRUDA®) in patients with advanced or metastatic solid tumors. The data set to be presented will include the full data from the monotherapy dose-escalation arm, updated data from the monotherapy dose-expansion arm and preliminary data from the combination study.
A photo accompanying this announcement is available at https://www.globenewswire.com/NewsRoom/AttachmentNg/d0b996ca-ed88-4a91-8a88-5e940c9cc44b
Notably, the MDNA11 abstract was submitted for presentation at the 2024 Annual Meeting of ASCO on 6th February 2024. On March 29th, 2024, the lead-author was notified by the ASCO Scientific Program Committee Chair that the submitted abstract was accepted as an oral podium presentation. On April 5th, 2024, AACR published the full abstracts for their 2024 Annual Meeting, including the MDNA11 abstract. On April 24th, 2024, 19 days after the publication of the AACR Abstracts, ASCO, and subsequently Medicenna, announced the acceptance of MDNA11’s abstract as an oral presentation. However, ASCO subsequently decided to withdraw the MDNA11 abstract from the ASCO 2024 program, citing “Prior Publication Policy Violation.”
Medicenna and the lead-author requested ASCO to reconsider their withdrawal explaining that while the data in the AACR 2024 abstract was time-limited and in-progress, the ASCO 2024 abstract covered the completed Phase 1 portion of the study and a good-faith intent to provide substantially new data never previously presented at any other conference, including AACR 2024 (see Table above). ASCO continues to maintain its position and the Company believes that it has fully exhausted all available avenues to secure a reversal of the decision by ASCO.
MDNA11 continues to demonstrate its potential to be a ‘best-in-class’ IL-2 agonist through its compelling single-agent activity and has been differentiated from other IL-2’s in clinical development by its deep and durable responses in the ABILITY-1 study amongst high-dose phase-2 eligible patients (N=14) with advanced solid tumors who have failed checkpoint inhibitor therapies. As of the March 22, 2024, data cut-off date, MDNA11 has demonstrated a response rate of 29% (4 partial responses) and a clinical benefit rate of 50% (4 partial responses, and 3 stable diseases > 24 weeks).
The second abstract of the Company, offering new data analyses for bizaxofusp (formerly known as MDNA55), a Phase-3 ready immunotherapy for recurrent glioblastoma, will be presented as a poster at the 2024 ASCO Annual Meeting, as previously announced by the Company. The full text of the published abstracts can be found on the 2024 ASCO Annual Meeting.
About MDNA11
MDNA11 is a long-acting ‘beta-enhanced not-alpha’ interleukin-2 (IL-2) Superkine specifically engineered to overcome the shortcomings of aldesleukin and other next generation IL-2 variants by preferentially activating immune effector cells (CD4+ T, CD8+ T and NK cells) responsible for killing cancer cells, with minimal or no stimulation of immunosuppressive Tregs. These unique proprietary features of the IL-2 Superkine have been achieved by incorporating seven specific mutations and genetically fusing it to a recombinant human albumin scaffold to improve the pharmacokinetic (PK) profile and pharmacological activity of MDNA11 due to albumin’s natural propensity to accumulate in highly vascularized sites, in particular tumor and tumor draining lymph nodes. MDNA11 is currently being evaluated in the Phase 1/2 ABILITY-1 study as both a monotherapy and in combination with pembrolizumab (KEYTRUDA®).
About the ABILITY-1 Study
The ABILITY-1 study (NCT05086692) is a global, multi-center, open-label study that assesses the safety, tolerability, pharmacokinetics, pharmacodynamics and anti-tumor activity of MDNA11 as monotherapy or in combination with pembrolizumab (KEYTRUDA®). In the combination dose escalation of the Phase 2 study, approximately 6-12 patients are expected to be enrolled and administered ascending doses of MDNA11 intravenously once every two weeks in combination with pembrolizumab. This portion of the study includes patients with a wide range of solid tumors with the potential for susceptibility to immune modulating therapeutics. Upon identification of an appropriate dose regimen for combination, the study will proceed to a combination dose expansion cohort.
About Medicenna
Medicenna is a clinical-stage immunotherapy company focused on developing novel, highly selective versions of IL-2, IL-4 and IL-13 Superkines and first-in-class Empowered Superkines. Medicenna’s long-acting IL-2 Superkine, MDNA11, is a next-generation IL-2 with superior affinity toward CD122 (IL-2 receptor beta) and no CD25 (IL-2 receptor alpha) binding, thereby preferentially stimulating cancer-killing effector T cells and NK cells. Medicenna’s IL-4 Empowered Superkine, bizaxofusp (formerly MDNA55), has been studied in 5 clinical trials enrolling over 130 patients, including a Phase 2b trial for recurrent GBM, the most common and uniformly fatal form of brain cancer. Bizaxofusp has obtained FastTrack and Orphan Drug status from the FDA and FDA/EMA, respectively. Medicenna’s early-stage BiSKITs™ (Bifunctional SuperKine ImmunoTherapies) and the T-MASK™ (Targeted Metalloprotease Activated SuperKine) programs are designed to enhance the ability of Superkines to treat immunologically “cold” tumors.
For more information, please visit www.medicenna.com, and follow us on Twitter and LinkedIn.
KEYTRUDA® is a registered trademark of Merck Sharp & Dohme LLC, a subsidiary of Merck & Co., Inc., Rahway, NJ, USA.
Forward-Looking Statements
This news release contains forward-looking statements within the meaning of applicable securities laws. Forward-looking statements include, but are not limited to, express or implied statements regarding the future operations of the Company, estimates, plans, strategic ambitions, partnership activities and opportunities, objectives, expectations, opinions, forecasts, projections, guidance, outlook or other statements that are not historical facts, such as statements on the Company’s clinical performance and potential of MDNA11 and bizaxofusp (MDNA55) as well as the statements relating to the event that the Company is planning to hold report on new clinical data from the ongoing Phase 1/2 ABILITY-1 Study evaluating MDNA11. Drug development and commercialization involve a high degree of risk, and only a small number of research and development programs result in commercialization of a product. Results in early-stage clinical studies may not be indicative of full results or results from later stage or larger scale clinical studies and do not ensure regulatory approval. You should not place undue reliance on these statements or the scientific data presented. Forward-looking statements are often identified by terms such as “will”, “may”, “should”, “anticipate”, “expect”, “believe”, “seek”, “potentially” and similar expressions. Forward-looking statements are based on a number of assumptions believed by the Company to be reasonable at the date of this news release. Although the Company believes that the expectations reflected in such forward-looking statements are reasonable, there can be no assurance that such statements will prove to be accurate. These statements are subject to certain risks and uncertainties and may be based on assumptions that could cause actual results and future events to differ materially from those anticipated or implied in such statements. Important factors that could cause actual results to differ materially from the Company’s expectations include the risks detailed in the latest Annual Report on Form 20-F of the Company and in other filings made by the Company with the applicable securities regulators from time to time in Canada.
The reader is cautioned that assumptions used in the preparation of any forward-looking information may prove to be incorrect. Events or circumstances may cause actual results to differ materially from those predicted, as a result of numerous known and unknown risks, uncertainties, and other factors, many of which are beyond the control of the Company. The reader is cautioned not to place undue reliance on any forward-looking information. Such information, although considered reasonable by management, may prove to be incorrect and actual results may differ materially from those anticipated or implied in forward-looking statements. Forward-looking statements contained in this news release are expressly qualified by this cautionary statement. The forward-looking statements contained in this news release are made as of the date hereof and except as required by law, we do not intend and do not assume any obligation to update or revise publicly any of the included forward-looking statements.
This news release contains hyperlinks to information that is not deemed to be incorporated by reference in this news release.
Investor and Media Contact:
Christina Cameron
Investor Relations, Medicenna Therapeutics
ir@medicenna.com
(647) 953-0673


