I came across an - article - about a year or so ago....
It descibed the interaction between ) sodium chloride brines + ammonia )
In short - it went on to say - sodium chloride would convert to - ammonia.
And here is where the smoking gun - could reveal much more about - blue + green copper + nickel hydroxide conversions.
It's not all just - chloride or sulphide.
Most ( laboratories ) will also use - nitric acid - beside the ususal - aqua regia.
Nitricpalys an important role in mineral conversions.
So.... if Sudbury basin all the way to upper Superior and the infamous feer belt,
down to Manatoulin and the limestones, to Great Lkaes and their numerous salt beds, and tying in Sudbury along with Lake Wanapetei - and all the chloric intrusions...
Let's now ask the questions -
what would be the carbonic acid + ammonia levels in - litho geology - specifically - the geology that overlays the diabade - which has seen significant chloric alteration.
Anyone up for - nitric + ammonia - assay - wink ?
Oxygen, Hydrogen, Nitrogen, Salt chlorides and the ammonia magic bullet -
Why magic bullet ?
Nitric acid does affect nickel bearing ores far quicker.
In Alberta -ammonia runs with in the - salt brines.
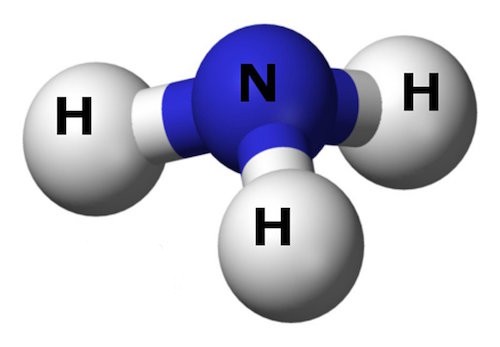
Ammonia molecule -
3 hydrogen + 1 nitrogen.
= nitrogen + dissolution mineral = bonded to = 3 hydrogen. ( water )
would fit like a glove - minerals converted to hydroxides due to sodic or chloric alterations with
nitric or ammonia presence.
.