Theralase Provides Update on 9 Months Post Treatment Cystoscopy Analysis
TORONTO, ONTARIO / ACCESSWIRE / November 8, 2018 / Theralase Technologies Inc.
("Theralase®" or the "Company")(TSXV: TLT) (OTCQB: TLTFF), a
clinical stagepharmaceutical company dedicated to the research and development of lightactivated Photo Dynamic Compounds
("PDCs") and their associated drugformulations intended to safely andeffectively destroy various cancers has
provided anupdate on patient five, enrolled and treated in the recently completed Phase IbNon-Muscle Invasive Bladder Cancer
("NMIBC")clinical study ("Study").
The Study's purpose was to evaluate TLD-1433,Theralase's lead PDC, for the primary endpoint of safety and tolerability, witha
secondary endpoint of pharmacokinetics (movement and exit of drug withintissue) and an exploratory endpoint of efficacy.
Theralase'sAnti-Cancer Treatment involves the instillation of a water-based solution of Theralase'slead anti-cancer PDC,
TLD-1433 at the Therapeutic Dose (0.70 mg/cm2), via a catheterinserted through the urethra into the bladder of the
patient, to allow the PDCto be preferentially absorbed by NMIBC tumours. The bladder is then drained ofthe solution, flushed with
sterile water to remove non-absorbed solution andrefilled with sterile water via a cystoscope. A fibre optic assembly, known asa
Laser Emitter emits laser light toactivate TLD-1433, while a proprietary Dosimetry
System detects the emitted laserlight, used for patient safety and efficacy. Both devices are inserted through
thecystoscope, with the sole purpose of activating the absorbed PDC to destroy theNMIBC tumours.
Thetreatment was well tolerated by the patient, who demonstrated no tumour recurrenceor presence of disease at the 90 day or 180
day clinical and cystoscopy assessment.
Thepatient has met Study endpoints demonstratingachievement of the primary, secondary and exploratory endpoints at 90 and
180days and now at 270 days post treatment that marks a new achievement for theCompany.
Arkady Mandel, M.D., Ph.D., D. Sc., Interim Chief Executive Officer andChief Scientific Officer of Theralase stated, "This is an
example of the enormous opportunity that awaits thisyoung Company in the treatment of cancer. For NMIBC, a Complete Response
("CR") is defined by the FDA as the definitiveendpoint for single-arm intravesical studies of patients who present
with BCG-UnresponsiveCarcinoma In-Situ ("CIS") disease,with or without resected papillary tumours. In the proposed
Phase II NMIBCclinical study, the Company is providing two treatment procedures (therapeutic atDay 0 and maintenance at Day 180).
The latest data on patient five is extremelyencouraging, in that it demonstrates after only one treatment procedure, CR at3, 6 and
now 9 months post-treatment has been obtained. If the efficacy resultsare able to be demonstrated at 12 months post-treatment, in a
larger patientpopulation, conducted in a well-designed Phase II NMIBC clinical study, thenthe Theralase Anti-Cancer Technology has
the potential to be the next goldstandard in the treatment of NMIBC. The Theralase Anti-Cancer Technology isalso multi-faceted, in
that the technology is able to be adapted to the treatmentof additional cancer indications, if successfully validated in
independent clinicalstudies. Pending successful commencement of the Phase II NMIBC clinical study, theCompany plans to investigate
the commencement of an additional Phase Ibclinical study for a new cancer indication."
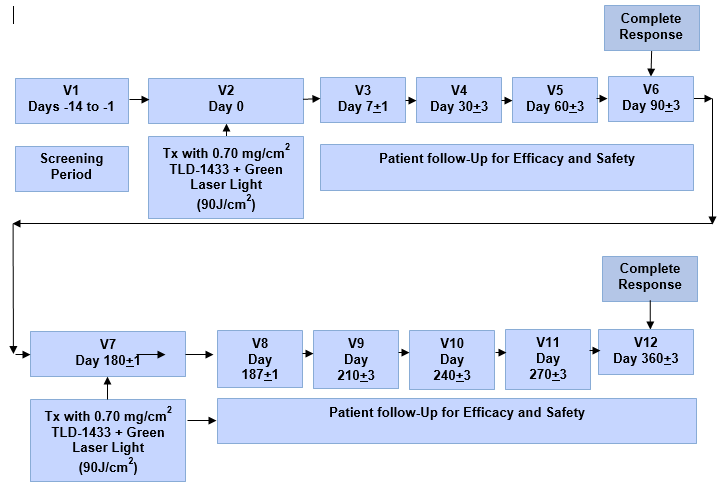
About the PhaseII NMIBC Clinical Study:
"A Phase II Clinical Study of Intravesical Photo Dynamic Therapy inPatients with BCG-Unresponsive Non-Muscle
Invasive Bladder Cancer or PatientsWho are Intolerant to BCG Therapy" will utilize the Therapeutic Dose(0.70 mg/cm2)
of TLD-1433 and will focus on the treatment ofapproximately 100 NMIBC patients in approximately 20 clinical sites located inCanada,
the US and internationally, with a primary endpoint of efficacy.
The endpointsof the Phase II NMIBC Clinical Study will be:
Primary(Efficacy) - Evaluated by Complete Response ("CR")in patients with Carcinoma In-Situ
("CIS") with or without resectedpapillary disease at 90 days post-treatment with duration of CR evaluated at360
days post-treatment.
PatientCR is defined as at least one of the following:
1)Negative cystoscopy and negative (including atypical) urine cytology
2)Positive cystoscopy with biopsy-proven benign or low-grade NMIBC
3)Negative cystoscopy with malignant urine cytology, if cancer is found in theupper tract or prostatic urethra and random bladder
biopsies are negative
Secondary (Safety) - Evaluatedby the incidence and severity of Adverse Events ("AEs") Grade 4
orhigher that do not resolve within 360 days post-treatment; whereby:
Grade 1= Mild, Grade 2 = Moderate, Grade 3 = Severe, Grade 4 = Life-threatening ordisabling and Grade 5 = Death
ProposedPhase II Clinical Study Treatment Plan:
About TheralaseTechnologies Inc.
Theralase® is a clinicalstage pharmaceutical company dedicated to the research and development of lightactivated Photo Dynamic
Compounds and their associated drug formulations intended to safely and effectively destroy various cancers.
Additional information isavailable at www.theralase.comand www.sedar.com.
This news release contains "forward-lookingstatements" which reflect the current expectations of management
of theCorporation's future growth, results of operations, performance and businessprospects and opportunities. Such statements
include, but are not limited to,statements regarding Theralase's proposed development plans with respect toPhoto Dynamic Compounds
and their drug formulations. Wherever possible, wordssuch as "may", "would",
"could","should", "will", "anticipate",
"believe","plan", "expect",
"intend","estimate", "potential for" and similarexpressions have been used to
identify these forward-looking statements. Thesestatements reflect management's current beliefs with respect to future eventsand
are based on information currently available to management. Forward-lookingstatements involve significant risks, uncertainties and
assumptions includingwith respect to the ability of Theralase to: successfully fund and complete
aPhase II NMIBC clinical study, secure the requisite regulatory approvals tocommence and fund a
Phase IINMIBC clinical study and implement itsdevelopment plans. Many factors
could cause the Corporation's actual results,performance or achievements to be materially different from any future
results,performance or achievements that may be expressed or implied by suchforward-looking statements; including, without
limitation, those listed in thefilings made by the Corporation with the Canadian securities regulatory authorities(which may be
viewed at www.sedar.com). Should one or more of these risks oruncertainties materialize or
should assumptions underlying the forward lookingstatements prove incorrect, actual results, performance or achievements mayvary
materially from those expressed or implied by the forward-lookingstatements contained in this news release. These factors should be
consideredcarefully and prospective investors should not place undue reliance on theforward-looking statements. Although the
forward-looking statements containedin the press release are based upon what management currently believes to bereasonable
assumptions, the Corporation cannot assure prospective investorsthat actual results, performance or achievements will be consistent
with theseforward-looking statements. The Corporation disclaims any intention orobligation to revise forward-looking statements
whether as a result of newinformation, future developments or otherwise except as required by law. Allforward-looking statements
are expressly qualified in their entirety by thiscautionary statement.
Neither TSX Venture Exchange nor itsRegulation Services Provider (as that term is defined in the policies of theTSX Venture
Exchanges) accepts responsibility for the adequacy or accuracy ofthis release.
For More Information:
1.866.THE.LASE (843-5273)
416.699.LASE (5273)
info@theralase.com
www.theralase.com
SOURCE: Theralase Technologies Inc.
